An implantable, remote-controlled device has provided a newer option for patients who fail sleep apnea therapies since it was introduced. One patient shares his story.
Obstructive sleep apnea (OSA) affects roughly 10-20 percent of Americans, causing sufferers to wake up tired and groggy. OSA can also significantly increase risk for serious long-term health problems. A newer treatment option of implanting a battery-powered device to open the airway may provide an effective option for people who have difficulties with conventional therapies, like continuous positive airway pressure (CPAP) therapy. Phillip Huyett, MD, a physician-surgeon in the Division of Sleep Medicine and Surgery at Mass Eye and Ear, explained how the device works and what to expect with surgery in an interview with Focus.
Untreated sleep apnea tied to major health woes
In OSA, the upper airway becomes intermittently blocked, limiting the amount of air that can reach the lungs during sleep. This reduces oxygen and disrupts sleep, raising risk for long-term health problems including heart attacks, heart failure, arrhythmias, strokes, high blood pressure, diabetes and early dementia.
OSA sufferers are typically diagnosed by a sleep study that measures their hourly sleep breathing disturbances, or Apnea Hypopnea Index (AHI). Severe sleep apnea is an AHI of 30 or higher.
The first-line therapy prescribed to affected individuals with moderate-severe OSA is CPAP. Each night, users wear a mask that is hooked up to a hose and a machine that delivers positive air pressure through the nose and/or mouth to open the airway. While it is the single most effective therapy, some studies show that up to 75 percent of people discontinue CPAP within the first 5 years of use.
Inability to tolerate CPAP may lead patients to consider second-line therapies. These include positional therapy, such as sleeping on your side or stomach, lifestyle changes like weight loss, oral appliances, and some surgical procedures. A newer therapy option, called hypoglossal nerve stimulation, has been used increasingly since it was approved by the FDA in 2014 for moderate to severe sleep apnea.
Remote-controlled device may be “game changer” for some
Hypoglossal nerve stimulation is an implantable, battery-powered device that is surgically placed under the skin above the chest. The device, called Inspire Upper Airway Stimulation (Inspire Medical Systems©), is connected to the nerve that controls tongue movement. Users turn on the device before bed with the use of remote control, otherwise there is no device on the face or in the mouth. During the night, the device moves the tongue slightly forward with each breath, relieving the obstructive event causing sleep apnea. Some studies have found this treatment can lead to a 79 percent reduction in sleep apnea events.
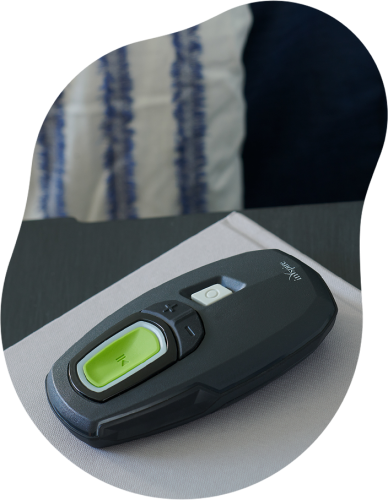
“This procedure can be a game-changer for some patients who have failed other therapies,” said Dr. Huyett. “In a perfect scenario, a patient will do well with CPAP, but unfortunately a subset do not. Then, we need to explore other options to prevent OSA from worsening a patient’s health and improve sleep quality.”
The implant surgery is done through three incisions in the skin and lasts about two to three hours. Patients go home the same day. After one month to allow the incisions to heal, Dr. Huyett turns on device to a minimal setting and teaches the patient how to use the remote. Then slowly over three months, the setting is increased until snoring and symptoms go away. A follow-up in lab sleep study is done where the device is fine-tuned, followed by an at-home sleep study three months later. The battery lasts about 11 years until it needs to be replaced. The device is adjustable and removable if a patient is not benefiting from therapy and/or decides it’s no longer for them. The surgical risks are minimal as is the post-operative pain; most patients use acetaminophen or ibuprofen.
Before considering the device or additional treatment options, patients must have tried and failed CPAP, then need to make an appointment with a sleep specialist to undergo further testing, such as a sleep endoscopy.
Severe sleep apnea case finally brought under control
No matter what treatment Dick Stagnone tried, he couldn’t get a sufficient night’s sleep.
The retired IT manager, father of two who now splits time between Massachusetts and Florida with his wife Paula, was first diagnosed with OSA in 1994. Back then, testing showed he had an extremely severe case of OSA with an AHI reading of 128.
His sleep apnea caused snoring, he woke with a foggy head and frequently felt sleepy.

Since that time he tried CPAP twice, failing both times within 30 days. He also tried a custom oral appliance, and underwent two surgeries: a nose surgery (septoplasty) and a soft-tissue procedure to remove the tonsils and tissues covering the back of the airway (uvulopalatopharyngoplasty, or UPPP). These surgeries helped his snoring, he said, but sleep tests still showed an AHI of 95 breathing disturbances per hour.
He started doing research soon after his diagnosis. He first came across the Inspire device in 2012 and was interested, but it was not FDA-approved at the time. Several years later when he came across community health talk hosted by Dr. Huyett on the device, he signed up. When he heard that the higher the AHI, the better a patient might respond to treatment, “that was all I needed to hear,” he told Focus. He emailed Dr. Huyett before the talk ended.
Mr. Stagnone underwent surgery in December 2019 and started using Inspire in January 2020. The device happened to be activated on the day of his 63rd birthday. His follow-up sleep study was delayed due to the COVID-19 pandemic, but he instead helped design a study where he measured his oxygen over night. His oxygen levels were previously in the mid-70th percentile during sleep each night and are now rarely below 90 percent. He has since undergone both an in-lab sleep study and a home sleep study. They demonstrated a dramatic improvement in his AHI, which is now down to about 12 per hour.
“I wake up without feeling a foggy head. I finally feel like I get an excellent night of sleep,” he said. “I’m a new man.”
Dr. Huyett is conducting ongoing research on patients using Inspire, including the study Mr. Stangone designed, to track its outcomes over a longer time. Future research will look at ways to predict which patients might benefit most from this therapy or other treatment options.
Visit our website for more information on hypoglossal nerve stimulation.
About Our Expert
 Dr. Phillip Huyett is a member of the Division of Sleep Medicine and Surgery at Mass Eye and Ear. He is fellowship trained in sleep medicine, and sees adult and pediatric patients at both of our Main Campus and Longwood locations in Boston.
Dr. Phillip Huyett is a member of the Division of Sleep Medicine and Surgery at Mass Eye and Ear. He is fellowship trained in sleep medicine, and sees adult and pediatric patients at both of our Main Campus and Longwood locations in Boston.
my doctor sent you a referral because i have severe sleep apnea, can someone please call me for appointment
Hi Frederick, please contact our sleep medicine division for an appointment at 617-573-3954. For more info: https://www.masseyeandear.org/specialties/sleep-medicine
Dr. Huyett started treating me a little over a year ago. After rounds and rounds of battling with my insurance company over whether they would approve the Inspire device (which they wouldn’t), we decided on the soft-tissue procedure to remove the tonsils and tissues covering the back of the airway (uvulopalatopharyngoplasty, or UPPP) as well as a couple others. I had my procedures in October and so far so good. I am sleeping and waking up not tired! When I follow back up with Dr. Huyett and have another sleep study we will determine if the Inspire is the next step in my treatment. I have lived with OSA for well over 10 years and I too fell prey to the CPAP eventually not working for me. I highly recommend reaching out and scheduling an appointment with Dr. Huyett. He is the best of the many sleep experts I have seen over the years and to date has given me my life back. Thank you.
Lenny, thanks so much for sharing your story and your comment, we’re so glad to hear you’re having better sleep. Thanks also for the comment on your experience with Dr. Huyett, so great to hear you had a good experience at Mass Eye and Ear.
I have been on the Sleep Apnea Machine twice. No one has told me how sever my apnea is. I heard 5 for the first time and 19 for the second time. I cannot get the adjustments made to reactivate the machine after I have toileted and i am either awake for the night or just go without. I am 86, presently in Florida and don’t seem to have a sleep problem. My cardiologist says it’s too bad I’m not on the machine. I don’t know whether I snore. I am very tired through the day. What do you say?
Hi Dorothy. Thanks for reading. We unfortunately can’t give medical advice over the blog. You can contact our sleep medicine division if you’re interested in setting up a virtual appointment. https://www.masseyeandear.org/specialties/sleep-medicine
I would like to proceed with an evaluation. Please contact me with the steps I need to follow to set up an appointment.
Thank you,
Cory Pierson