Two new studies may shed light on how patients with vestibular schwannoma (also known as acoustic neuroma), a type of intracranial tumor, may fare with their hearing loss prognosis and response to treatments.
Two studies published this past month provide new information about the tough-to-treat tumors, and motivate a future wave of studies tracking patients in an effort to better predict their outcomes, according to their lead study author, Konstantina M. Stankovic, MD, PhD, director of the Division of Otology and Neurotology at Mass Eye and Ear.
“There is currently no way to robustly predict whether a vestibular schwannoma will be tightly adherent to the nearby facial nerve, possibly resulting in incomplete tumor removal or facial weakness after surgery,” said Dr. Stankovic, the Sheldon and Dorothea Buckler Chair in Otolaryngology–Head and Neck Surgery at Mass Eye and Ear. Our identification of a new blood biomarker and increased understanding of how tumor-secreted molecules might affect hearing loss motivates us to perform more research, in the hopes of one day developing more targeted prognostic tests and treatments for vestibular schwannoma patients.”
Vestibular schwannoma may cause hearing loss in unaffected ear
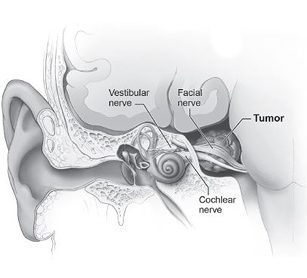
Vestibular schwannoma, also known as an acoustic neuroma, is a noncancerous tumor that arises from a balance (vestibular) nerve within the skull, adjacent to the hearing and facial nerves. Based on tumor biology and nerves it compresses, vestibular schwannoma can lead to hearing loss, tinnitus, balance problems, and additional effects on function including facial paralysis. If the tumor compresses the brain stem, it can cause major problems such as hydrocephalus (a buildup of fluid in the brain), and in rare cases, death.
Sensorineural hearing loss (SNHL) is the most common complaint in patients with this type of tumor. This form of hearing loss occurs not only from a tumor compressing the auditory nerve but also from tumor-secreted molecules. If these molecules are able to filter through the fluid that surrounds the brain or blood, then they may affect hearing in the opposite, non-tumor-affected side as well.
In the new study published online August 18 in Frontiers in Neurology. Dr. Stankovic and colleagues identified a higher risk of hearing loss in the unaffected ear.
Researchers carefully looked through a Mass Eye and Ear database of hearing tests to track long-term hearing outcomes in 661 patients with vestibular schwannoma in one ear. They found that compared to healthy people of the same age and sex, patients with vestibular schwannoma were significantly more likely to have the opposite ear affected by moderate SNHL.
This was the largest study to date quantitatively tracking hearing on both sides in patients with vestibular schwannoma, with the longest follow-up period of up to 24 years. The researchers urge clinicians to counsel people with these tumors to make sure to protect their hearing in their unaffected ear as well.
Addressing difficulties predicting how patients will respond to treatments
Unfortunately, it is unclear why some patients respond better to existing treatments for vestibular schwannoma and why others have poor outcomes following treatment due to limited data.
There are four management options for vestibular schwannoma: observing the tumor without treating if it is not growing, removing the tumor surgically, radiating the tumor, or entering a clinical trial to test an experimental drug that may stall the tumor’s growth. Each of these options carries risks, including worsening SNHL, tinnitus, dizziness and facial paralysis.

Currently the decision on how to treat vestibular schwannoma factors in the size of the tumor, its growth rate and where along a vestibular nerve it is located. There are no drugs FDA-approved for vestibular schwannoma or the associated SNHL, making surgery and radiation the most common treatment options.
This uncertainty is what led the researchers to look for biomarkers, or predictive markers in the blood, that may help determine which patients may be at greater risk from surgical tumor removal.
Comparing previous research with patients’ tumor tissue and blood yields potential testing target
To identify a biomarker, the researchers first utilized a previous study in which they performed the largest analysis to date of genes abnormally active in vestibular schwannoma. They then cross-referenced those findings with a database of protease enzymes (which break down proteins in the body) that are known to play an important role in tumor growths in cancer. There the researchers found one compound that stood out, called matrix metalloprotease 14 or MMP-14.
The researchers further explored that finding by looking at tumor tissue samples surgically collected from more than 20 patients. They found that MMP-14 was abundantly present in these tumors.
The researchers then developed a new test to detect activity of MMP-14 enzyme in patients’ blood that was collected at the time of surgery and prior to the removal of the tumor, and in their tumor secretions from specimens removed during surgery.
They found that those with higher amounts and activity of MMP-14 in the blood and tumor secretions were more likely to have worse tumor-induced SNHL and poorer surgical outcomes. Their findings were published July 28 in Frontiers in Molecular Neuroscience.
The researchers hope to conduct more studies by tracking patients with vestibular schwannoma over long periods of time to better see how their levels of MMP-14 correlate with their outcomes, in the hopes of one day developing a prognostic test and a targeted therapeutic for all patients with this type of tumor.
In addition to Dr. Stankovic, authors of the contralateral hearing loss paper include Samuel Early, Charlotte E. Rinnooy Kan, Maura Eggink, and Johan H. M. Frijns.
Co-authors of the MMP-14 paper include Yin Ren, Hiroshi Hyakusoku, Jessica E. Sagers, Lukas D. Landegger, and D. Bradley Welling.
Funding for these research projects was supported by National Institute on Deafness and Other Communication Disorders (R01DC015824), Nancy Sayles Day Foundation, the Lauer Tinnitus Research Center, the Zwanziger Foundation, the Barnes Foundation, and Shelley and Dorothea Buckler.

I have had two AN tumors, 2001 (removed) and 2020 (radiation)both on the Left side. Had a hearing test in 2004ish and another in November 2020 and results were no hearing loss in my good right ear.